Larks
Posts 28
Comments1650
Topic contributions4
Thanks for sharing this, it was a very interesting read!
I do want to question this claim though:
"But the alternative to a project like the AMA is that essential HIV treatments arrive half a decade late in places that needed them most."
It seems to me that an attractive alternative would be for African countries to simply give up on doing their own drug approvals, and outsource the decision making entirely to the FDA, MHRA, PMDA, EMA etc. Why not simply say that any drug approved by any of these agencies is automatically approved? This way drugs would be approved swiftly and with almost zero cost to both government and corporation.
You discuss institutions, but I don't think you discuss the right kind of institutions. If I am comparing Malawi to some of the other nearby landlocked African countries you mention, the first thing that jumps out to me is their dramatically worse economic freedom. Malawi has one of the worst scores in the entire world, ranked 147/165, a level more typical of central or saharan african countries.
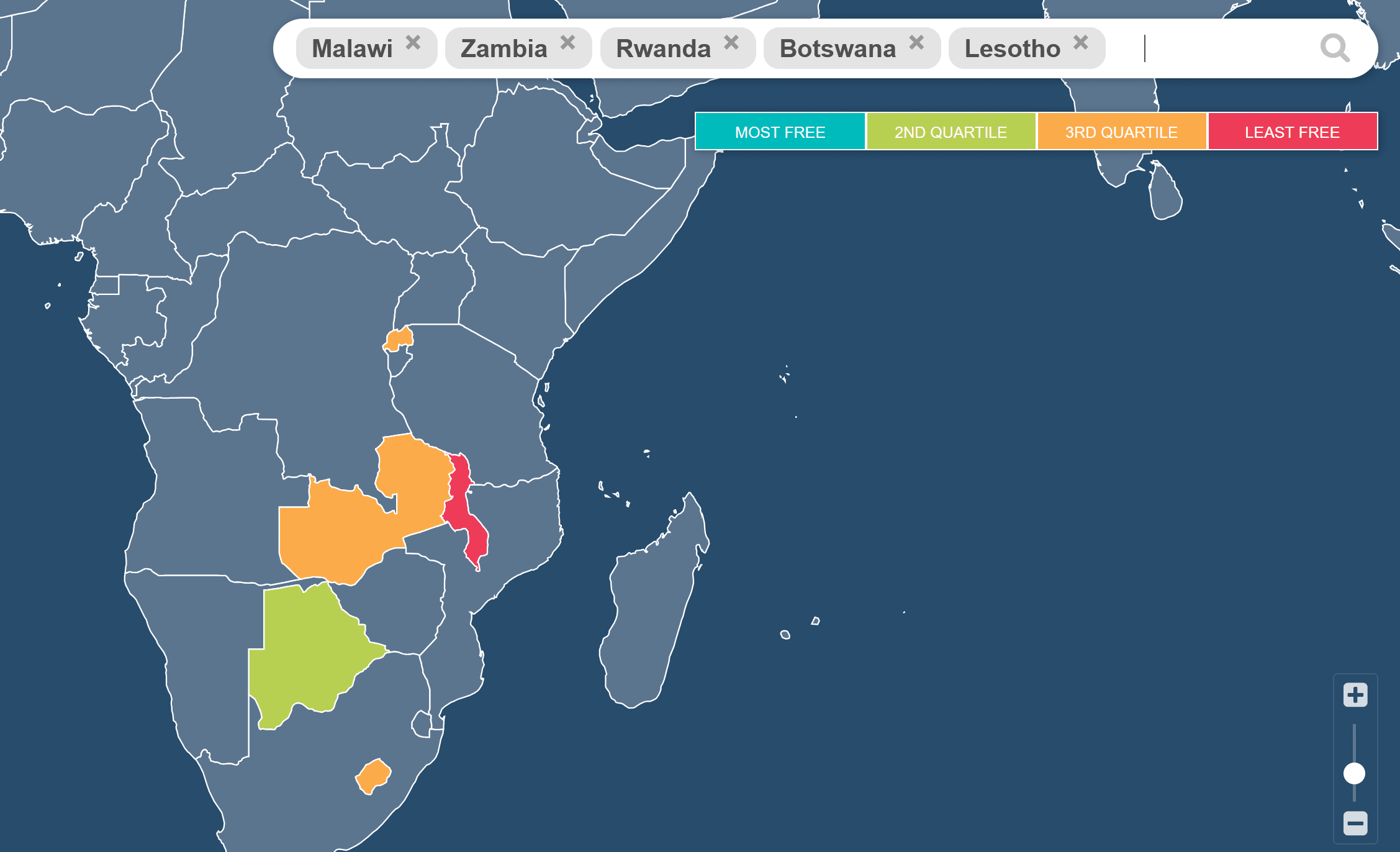
https://www.efotw.org/?geozone=world&page=map&year=2023&countries=MWI,ZMB,RWA,BWA,LSO#country-info
(You didn't mention Eswatini or Burundi, but they also score very badly - unfortunately the map tool above will only let me display 5 countries so I focused on those named in the text plus Zambia as it is neighbouring).
This doesn't resolve the infinite-regress style question of what causes some countries to have more capitalist institutions than others, but when it comes to which institutions to investigate, I think it is their economic institutions we should focus on.
If you don't gate access to your research resources (grant funding, GPUs etc.), and you have a non-trivial amount to give away, then I would expect almost all of them will end up going to projects you would not approve of.
Note that UBI does gate access - it gates to a (small) finite quantity per person.
Thanks for this very interesting article.
One quick suggestion: assuming that 100% of distributed cups are used seems quite aggressive to me. Using a 7.5 year average life I think suggests you are implicitly selecting on very enthusiastic adopters; I would imagine a lot are distributed and then discarded.

Thanks for replying!